THE CONFORMATION OF BUTANE
(C4H10)

PART I: The Newman Projections of Butane
The illustration of butane above is represented by the Newman Projections below by designating the
two middle carbons, one as the "front" and one as the "behind" carbon, and connecting the two end methyl groups accordingly.
PART II: Comparing Potential Energies Based on the Dihedral Angle
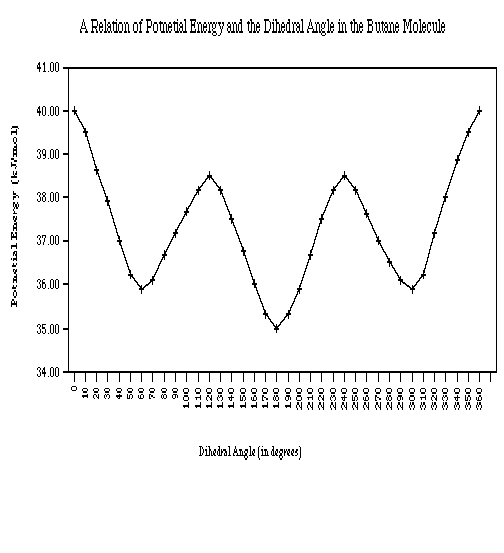
| Dihedral Angle (in degrees) | Conformation | kcal/mol difference from 0° |
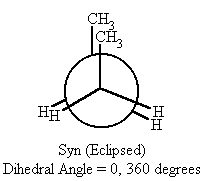
| 0 and 360 | syn (eclipsed) | none |
|---|
| 30 | no name | -2.0 |
|---|
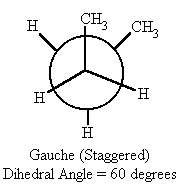
| 60 | gauche (staggered) | -4.1 |
|---|
| 90 | no name | -2.8 |
|---|
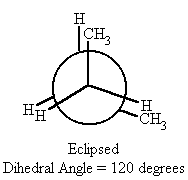
| 120 | eclipsed | -1.5 |
|---|
| 150 | no name | -3.3 |
|---|
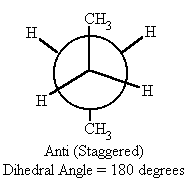
| 180 | anti (staggered) | -5.0 |
|---|
| 210 | no name | -3.3 |
|---|
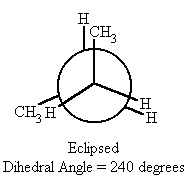
| 240 | eclipsed | -1.5 |
|---|
| 270 | no name | -2.8 |
|---|
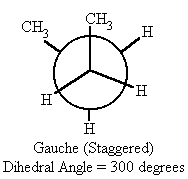
| 300 | gauche (staggered) | -4.1 |
|---|
| 330 | no name | -2.0 |
|---|
- When the dihedral angle = 0° or 360°, (syn conformation) in butane, the two methyl groups are in close proximity with the molecule, therefore the potential energy is at its highest.
- When the dihedral angle = 60° or 300°, (gauche conformation) in butane, the methyl groups are farther apart and therefore the potential energy drops by 4.1 kcal/mol.
- When the dihedral angle = 120° or 240°, (eclipsed conformation) in butane, the methyl groups align themselves with hydrogens on the same plane, resulting in the butane having a higher potential energy than the gauche conformations though still having a lower potential energy than the syn conformation ( dihedral angle = 0° or 360°) by 1.5 kcal/mol.
- In the anti conformation (when the dihedral angle = 180°) the potential energy is at its lowest, therefore the most stable. This is due to the methyl groups being the farthest away from each other in the molecule.
GRAPH: The Potential Energy of Butane as a Function of the Dihedral Angle
MOVIE: The Relationship of the Dihedral Angle and
the Potential Energy of Butane Reprised
Click to download the movie (714k)
Bryan Modders
with Dr. Abby Parrill
Department of Chemistry
Michigan State University
These pages may be downloaded and linked from other pages freely for academic and educational purposes. Questions, problems, and errors should be sent to
parrill@argus.cem.msu.edu
Original pages can be found here, local version by Lionel Delaude, 22 october 1997.