"Imidazol(in)ium-2-carboxylates as N-Heterocyclic Carbene Precursors in Ruthenium-Arene Catalysts for Olefin Metathesis and Cyclopropanation"
Adriana Tudose, Albert Demonceau, and Lionel Delaude
 |
source: Journal of Organometallic Chemistry
year: 2006
volume: 691
first page: 5356
last page: 5365
doi: 10.1016/j.jorganchem.2006.07.035
|
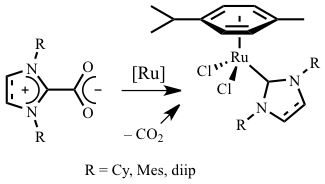
Abstract: Five imidazol(in)ium-2-carboxylates bearing cyclohexyl, mesityl, or 2,6-diisopropylphenyl substituents on their nitrogen atoms were prepared from the corresponding N-heterocyclic carbenes (NHCs) by reaction with carbon dioxide. They were characterized by IR and NMR spectroscopies, and by TGA. Their ability to act as NHC precursors for in situ catalytic applications was probed in ruthenium-promoted olefin metathesis and cyclopropanation reactions. When visible light induced ring-opening metathesis polymerization of cyclooctene or cyclopropanation of styrene with ethyl diazoacetate were carried out at 60 °C in the presence of [RuCl2(p-cymene)]2, the NHC.CO2 adducts and their NHC.HX counterparts (X = Cl, BF4) displayed similar activities. When metathesis polymerizations were performed at room temperature, the carboxylates proved far superior to the corresponding imidazol(in)ium acid salts. They displayed the same level of activity as the preformed RuCl2(p-cymene)(IMes) complex, whereas the combination of NHC.HX and KO-t-Bu were almost totally inactive. Results obtained for cyclopropanation reactions at room temperature did not show such a large discrepancy of behavior between the two types of adducts.
Keywords: Cyclooctene, Styrene, Ring-Opening Metathesis Polymerization, Cyclopropanation, Homogeneous Catalysis
[Full Text] [<< Previous Article] [Back to the List of Publications] [Next Article >>] l.delaude@ulg.ac.be