"The Chemistry of Azolium-Carboxylate Zwitterions and Related Compounds: a Survey of the Years 2009-2020"
Lionel Delaude
 |
source: Advanced Synthesis & Catalysis
year: 2020
volume: 362
first page: 3259
last page: 3310
doi: 10.1002/adsc.202000639
|
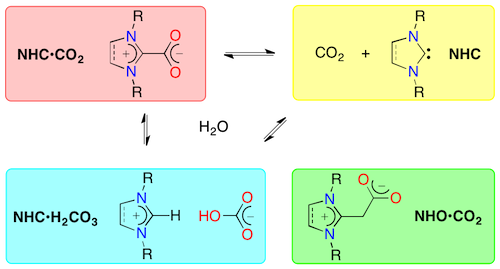
Abstract: This review highlights the most significant advances that were accomplished in the chemistry of azolium-carboxylate betaines within the years 2009-2020. Whenever needed, prior, seminal achievements are also briefly recalled to help put recent developments in a broader perspective. The compounds under scrutiny are zwitterions obtained upon nucleophilic addition of N-heterocyclic carbenes (NHCs) onto carbon dioxide. We first present an overview of the various synthetic paths that give access to these inner salts before discussing their stability. The main part of this report then summarizes the numerous applications relying on NHC·CO2 zwitterions employed either in stoichiometric or catalytic proportions in the fields of organometallic chemistry and homogeneous catalysis, organic synthesis and organocatalysis, polymer chemistry, and materials science. A particular emphasis is placed on the endeavors devoted to trap carbon dioxide and to convert this greenhouse gas into valuable chemicals via the intermediacy of NHC·CO2 zwitterions. Whenever available, mechanistic considerations that help assess the pivotal role played by these species in CO2 activation are discussed. Because of their close relationship with azolium-carboxylate betaines, azolium hydrogencarbonate salts and azolium-methylenecarboxylate zwitterions resulting from the addition of carbon dioxide onto N-heterocyclic olefins (NHOs) are included in this survey, which also covers the reactivity of carbon monoxide toward NHCs and related carbene species.
Keywords: Azolium Hydrogencarbonates, Carbon Dioxide Fixation, Green Chemistry, N-Heterocyclic Carbenes, N-Heterocyclic Olefins, Organocatalysis, Polymerization
[Full Text] [<< Previous Article] [Back to the List of Publications] [Next Article >>] l.delaude@ulg.ac.be