"Pd-PEPPSI Catalysts Bearing N-Heterocyclic Carbene Ligands Derived from Caffeine and Theophylline for Mizoroki-Heck and C(sp2)-H Arylation Reactions"
François Mazars, Koffi Sénam Etsè, Guillermo Zaragoza, and Lionel Delaude
 |
source: Journal of Organometallic Chemistry
year: 2023
volume: 1003
article: 122928
pages: 10
doi: 10.1016/j.jorganchem.2023.122928
|
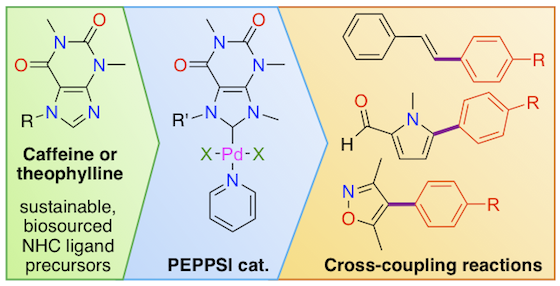
Abstract: The activity of three Pd-PEPPSI catalyst precursors bearing N-heterocyclic carbene (NHC) ligands derived from caffeine and theophylline was tested in Mizoroki-Heck reactions between styrene and five different aryl bromides bearing electron-donating or withdrawing substituents. Moderate to excellent conversions (44-98%) were achieved within 3 h at 100 °C with K2CO3 as the base and 1 mol% of catalyst in a green water/isopropanol mixture under a normal atmosphere. Electron-withdrawing groups favored the reaction and led to the highest yields of stilbene products (up to 85% after chromatographic purification). The activity of the three [PdX2(NHC)(Py)] complexes (X = Cl, I) was also probed in the C(sp2)-H arylation of two heterocyclic substrates using the same set of five representative aryl bromides. Experimental conditions were further optimized on a model reaction (1 mol% of catalyst in DMA at 120 °C for 4 h with KOAc as the base). Gratifyingly, 1 methyl-2-pyrrolecarboxaldehyde afforded high conversions (59-99%) and yields of isolated products (49-85%), with the best results obtained again under the influence of electron-withdrawing substituents. When a more challenging substrate, viz., 3,5-dimethylisoxazole was used, modest to satisfactory conversions (41-80%) and yields (30-69%) could still be reached with the best catalyst for each reaction. The structural properties of the complexes under investigation were compared using X-ray crystallography and Hirshfeld surface analysis.
Keywords: Alkaloids, Carbene Ligands, Cross-Coupling, Green Chemistry, Hirshfeld Surface
[Full Text] [<< Previous Article] [Back to the List of Publications] [Next Article >>] l.delaude@ulg.ac.be